
The clinical management of metabolic disorders is undergoing a radical shift as researchers move beyond generic tissue analysis to decode the intricate molecular mechanisms governing individual pancreatic cells. Historically, the medical community viewed type 2 diabetes through the lens of

For patients living with a chronic Achilles rupture that has resisted months of therapy and bracing, every step can feel like a wager against pain, weakness, instability, and the fear that a compromised tendon will fail again during even the simplest tasks. Traditional reconstruction can fix the
The pharmaceutical landscape is currently witnessing a seismic shift as Eli Lilly commits a staggering seven billion dollars to acquire Kelonia Therapeutics, a move that signals a departure from traditional oncology treatments toward advanced genetic engineering. This acquisition, which includes an

The high-stakes world of oncology research is currently undergoing a structural transformation as the 2026 grant cycle shifts away from traditional paradigms toward a more integrated, technology-heavy framework. This movement is not merely a change in administrative preference but a response to the
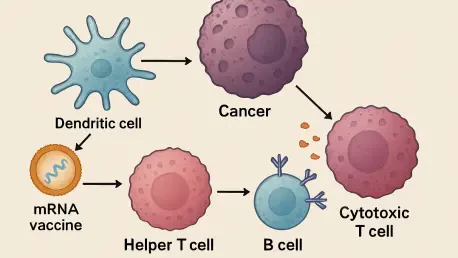
The sophisticated machinery of the human immune system often operates with a level of redundancy that ensures survival even when primary defenses are compromised by malignant growth. While the global success of messenger RNA technology was initially demonstrated through the rapid development of
The chronic burden of managing insulin-dependent diabetes has historically demanded a relentless cycle of blood glucose monitoring and hormonal injections that rarely achieve perfect physiological balance. Despite significant advancements in insulin delivery systems and continuous glucose monitors,