
As a leading figure in biopharma innovation and research, Ivan Kairatov has spent years navigating

The subtle architecture of a brain tumor often conceals a lethal potential that remains invisible

The launch of the Food and Drug Administration's Real-Time Clinical Trials pilot initiative during the current fiscal cycle marks a significant step toward updating a drug development process often criticized for being slow and technologically outdated. This move signals a departure from the

The integrity of global health security rests upon the unwavering commitment of scientists to follow the strict legal and safety protocols designed to prevent the accidental or intentional spread of dangerous pathogens; however, recent events have demonstrated that even high-level experts are not

The ability to monitor the precise journey of therapeutic cells within the human body has remained
In the rapidly evolving landscape of oncology, the ability to decode the complex conversations

The biopharmaceutical sector is entering a new era. After last year's market disruptions, the industry faces a harsh truth: scientific breakthroughs alone are no longer enough to keep ahead of the…

In 2026, boards and customers are asking a direct question: can promising science be turned into

The pharmaceutical sector is entering a decisive phase as obesity therapeutics gain more ground as
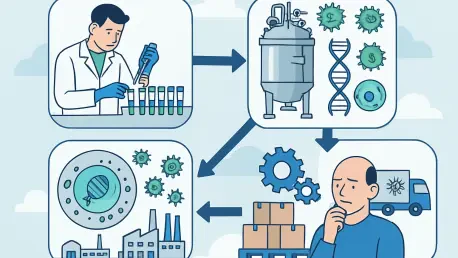
Advanced Therapy Medicinal Products cannot be scaled the same way biologics were. The core

The End of the Old Biopharma Playbook The biopharmaceutical industry is confronting a structural

The traditional method of identifying unknown chemicals in medical devices has relied on a precarious game of structural "look-alikes" that often misses the mark by an order of magnitude. In pharmaceutical and medical device manufacturing, ensuring that products are free from harmful Extractables

Healthcare systems have long struggled to provide equitable preventative services to patients

The traditional medical model for managing respiratory oncology has long relied on late-stage