
Recent research, published in the Journal of Translational Internal Medicine, delves into the intriguing association between gastroesophageal reflux disease (GERD) and cardiovascular health. GERD, primarily recognized as a digestive disorder characterized by acid reflux and heartburn, has now been

A groundbreaking study led by researchers from The University of Texas at Austin and Harvard Medical School has challenged the prevailing assumption that infants exposed to gestational diabetes mellitus (GDM) are at an automatically higher risk of developing childhood obesity. Published in the

In the quest to improve the quality of life for the aging population, a research team from Kyushu University has made a groundbreaking discovery that may pave the way for combating age-related muscle atrophy. As people age, the decline in muscle mass and function becomes a significant concern,

The intricate dance between viruses and the host cells they infect has always been a subject of intense scientific inquiry. Recent research into SARS-CoV-2, the virus at the heart of the COVID-19 pandemic, has revealed a sophisticated mechanism through which this pathogen manipulates host

Polycystic Ovary Syndrome (PCOS) is a common hormonal disorder affecting women of reproductive age. Characterized by symptoms such as irregular menstrual cycles, excessive androgen levels, and ovarian cysts, PCOS is also associated with significant metabolic complications like insulin resistance,
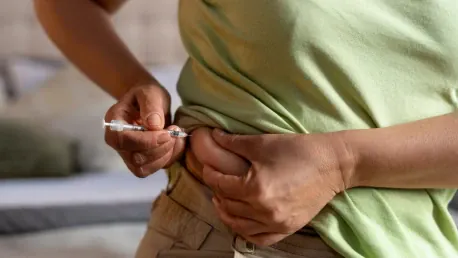
The eligibility criteria for semaglutide, a medication that has seen rapidly expanded use beyond its initial approval for type 2 diabetes, reflect broader implications for healthcare policies and pharmaceutical budgets in the United States. Initially, semaglutide was approved by the US Food and