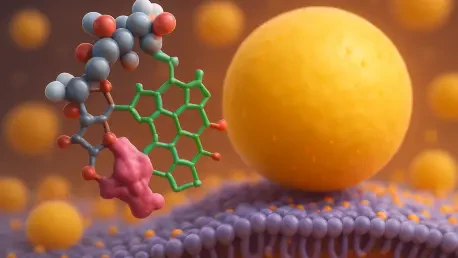
Scientists have finally pioneered a way to smuggle potent metabolic medicine directly into cells, effectively bypassing the dangerous systemic side effects that have historically stalled pharmaceutical progress. This breakthrough centers on a sophisticated “Trojan horse” delivery system that combines the appetite-suppressing power of modern incretin therapies with the deep metabolic benefits of insulin-sensitizing agents. By focusing on precision rather than broad circulation, this research offers a potential solution for patients who require more intensive treatment than current medications provide. This article explores the development of this hybrid molecule and analyzes how its targeted approach could redefine the future of metabolic health.
The objective of this exploration is to answer the most pressing questions regarding how this multi-targeted therapy works and why it represents a significant leap forward. Readers can expect to learn about the specific chemical mechanisms that allow the drug to enter cells and the preclinical evidence supporting its safety and efficacy. By understanding the limitations of existing treatments, one can better appreciate the innovation behind this quintuple-action molecule. The scope of this discussion covers the transition from systemic administration toward a more localized, cell-specific strategy that targets the root causes of obesity and type 2 diabetes.
Key Questions or Key Topics Section
Why Are Conventional Obesity Medications Limited by Systemic Side Effects?
The current landscape of metabolic medicine is dominated by incretin-based drugs, such as GLP-1 and GIP receptor agonists, which have demonstrated remarkable success in suppressing appetite and managing blood sugar. However, for many patients with severe insulin resistance, these hormones alone are not always sufficient to achieve optimal health outcomes. Physicians have long sought to incorporate insulin sensitizers, particularly a class of drugs known as PPAR agonists, which can fundamentally improve how the body processes energy. Despite their potency, these drugs have historically been difficult to use because they interact with receptors throughout the entire body, leading to unintended consequences.
When these sensitizers circulate freely in the bloodstream, they often affect tissues they were never meant to target. This lack of precision frequently results in severe side effects, such as significant fluid retention, anemia, and paradoxically, weight gain in the form of increased fat storage. Because the risks often outweighed the rewards, many promising compounds remained underutilized or were restricted to low, less effective doses. Consequently, the medical community faced a persistent challenge: how to harness the metabolic benefits of these powerful agents without triggering a cascade of systemic toxicity.
How Does the Hybrid Trojan Horse Mechanism Target Specific Cells?
The innovation introduced by the research team involves chemically linking an incretin component to a second pharmacological agent called lanifibranor. This design functions like a “Trojan horse,” where the incretin portion serves as the address label that identifies specific target cells. Because receptors for GLP-1 and GIP are primarily located on cells involved in metabolic regulation, the hybrid molecule ignores most other tissues. This allows the medication to concentrate exactly where it is needed most, significantly reducing the amount of drug required to achieve a therapeutic effect.
Once the incretin part of the molecule binds to the receptor on the cell surface, the entire complex is pulled inside the cell. Within this protected environment, the “cargo”—the lanifibranor—is released to interact with the cell nucleus. This internal release is critical because it activates three distinct nuclear receptors known as PPAR alpha, gamma, and delta. By combining these with the two surface receptors, the single molecule achieves a quintuple-action effect. This localized activation ensures that the potent metabolic switches are toggled only within the designated cells, leaving the rest of the body unaffected by the drug’s more aggressive components.
What Metabolic Improvements Were Observed During Preclinical Testing?
During laboratory trials involving models of diet-induced obesity, the hybrid molecule consistently outperformed traditional single-target or dual-target therapies. The data revealed that the subjects treated with this new compound experienced more significant weight loss and a sharper reduction in food intake compared to those receiving standard GLP-1 or GIP treatments. Moreover, the hybrid drug demonstrated a superior ability to stabilize blood glucose. It achieved this by enhancing the movement of sugar from the blood into the tissues while simultaneously signaling the liver to stop overproducing glucose, creating a comprehensive dual-action management system.
The most promising aspect of the research was the virtual disappearance of traditional side effects. Despite the inclusion of a PPAR agonist, there was no evidence of the anemia or fluid retention that typically plagues this class of medication. Furthermore, the researchers noted improvements in markers of liver and heart health, suggesting that the treatment offered protective benefits beyond simple weight management. While gastrointestinal symptoms remained similar to those seen with existing incretin therapies, the overall safety profile was dramatically improved, proving that cell-specific delivery could finally unlock the full potential of high-potency metabolic drugs.
Summary or Recap
The development of this hybrid molecule signaled a paradigm shift in how metabolic disorders are addressed, moving from broad systemic intervention toward microscopic precision. By activating five different drug targets through a single delivery vehicle, the research demonstrated that it was possible to maximize metabolic benefits while nearly eliminating the toxicities that once limited pharmaceutical options. This quintuple-action approach not only enhanced weight loss and glucose control but also offered a protective effect on vital organs like the heart and liver. The success of the “Trojan horse” strategy provided a blueprint for future drug design, emphasizing that the delivery method is just as important as the chemical payload itself.
This unified strategy offered a safer alternative for patients who might have previously been at risk for complications from systemic sensitizers. The findings reinforced the importance of incretin activity as a gateway for other therapeutic agents, allowing researchers to toggle complex metabolic switches with unprecedented accuracy. For those interested in the molecular specifics, the study highlighted how specific receptor binding could effectively isolate pharmacological activity. As the medical community looks toward more personalized and effective treatments, this hybrid model stands as a testament to the power of intelligent molecular engineering in overcoming long-standing medical hurdles.
Conclusion or Final Thoughts
The researchers successfully proved that the limitations of traditional drug therapy were not necessarily due to the drugs themselves, but rather the way they were distributed throughout the body. By refining the delivery mechanism, the team opened a door that had been closed for decades, allowing for the use of potent sensitizers in a controlled, safe manner. This achievement prompted a broader reflection on how other dangerous but effective compounds might be repurposed through similar targeted strategies. It underscored the reality that the next generation of medicine would likely rely on these multifaceted, hybrid structures to treat complex diseases.
Looking forward, the transition of this therapy from preclinical models into human clinical trials became the primary focus for developers. Because human biology presents unique challenges, particularly regarding how certain receptors function, the molecule required further optimization to ensure the same success seen in early trials. Establishing strong partnerships between academic institutions and the pharmaceutical industry was identified as the essential next step to bring this innovation to the patient’s bedside. Ultimately, the successful creation of this targeted molecule provided a hopeful trajectory for the treatment of obesity, suggesting that the most difficult metabolic challenges might soon be manageable through precision science.