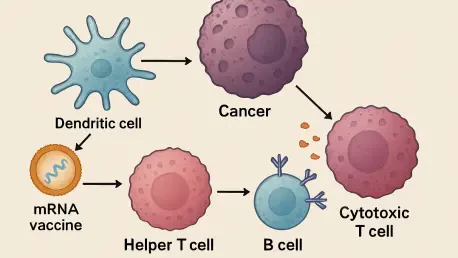
The sophisticated machinery of the human immune system often operates with a level of redundancy that ensures survival even when primary defenses are compromised by malignant growth. While the global success of messenger RNA technology was initially demonstrated through the rapid development of viral vaccines, its application in the field of oncology has required a much deeper understanding of internal biological interactions. Researchers have recently focused on how these vaccines communicate with the body’s complex network of immune cells to trigger a specific and aggressive response against tumors. This investigation into the underlying mechanics of immunotherapy provides a clearer picture of why certain treatments succeed where others fail, moving the scientific community closer to highly personalized medical interventions.
The objective of this analysis is to explore the revolutionary discovery of dual immune pathways that mRNA vaccines utilize to combat cancer. Readers can expect to learn about the specific roles of various dendritic cell subtypes and how their interaction with T cells determines the effectiveness of a vaccine. By examining the results of groundbreaking studies conducted at institutions like Washington University School of Medicine, this article sheds light on the “backup systems” of the immune system that were previously overlooked. The scope of the content includes the transition from traditional immunological theories to a new model that accounts for the versatility and resilience of mRNA-based therapies in the current landscape of 2026.
Key Questions Regarding Dual Immune Pathways
Why Is the Identification of Multiple Dendritic Cell Pathways Significant for Oncology?
For a significant period, the prevailing consensus among immunologists suggested that a single type of cell was responsible for the success of cancer vaccines. This “gatekeeper” was known as the classical type 1 dendritic cell, or cDC1, which was thought to be the only vehicle capable of “teaching” killer T cells how to identify and destroy malignant tissue. Because dendritic cells act as messengers that present protein fragments to the rest of the immune system, scientists believed that if the cDC1 pathway was damaged or suppressed by cancer, the vaccine would inevitably fail to produce a response. This singular focus limited the development of treatments for patients whose immune systems did not follow this specific blueprint.
However, recent research has dismantled this narrow view by demonstrating that mRNA vaccines are far more adaptable than previously imagined. When scientists observed mouse models that lacked the cDC1 cell type, they were surprised to find that the mRNA vaccines remained highly effective at clearing tumors. This revealed the existence of a parallel pathway that the immune system can activate when the primary route is unavailable. The significance of this finding cannot be overstated, as it suggests that mRNA technology possesses a built-in redundancy that allows it to work across a broader range of biological profiles, making it a more reliable tool for a diverse patient population.
How Does the Cross-Dressing Mechanism Enhance the Efficacy of mRNA Vaccines?
The discovery of a second pathway led researchers to focus on the classical type 2 dendritic cell, or cDC2, which was typically associated with different types of immune responses. While cDC1 cells are known for their internal processing of proteins, cDC2 cells utilize a unique and somewhat unconventional method to gather information about threats. This method is referred to as “cross-dressing,” a process where the dendritic cell does not necessarily produce the tumor-specific proteins itself. Instead, it acquires pre-processed protein fragments and membrane complexes from other cells that have already translated the vaccine’s instructions.
This outsourcing of information allows the cDC2 cells to become effective instructors for killer T cells even without direct interaction with the mRNA particles. By effectively “wearing” the markers of the tumor provided by other cells, these dendritic cells can stimulate a robust and aggressive immune response. This mechanism acts as a force multiplier for the vaccine, ensuring that the message to attack the cancer is spread throughout the immune system through multiple channels. The ability of the immune system to use these two distinct methods of “education” explains the high potency of mRNA vaccines compared to older, more traditional vaccine platforms that rely on a single mode of activation.
What Role Do Molecular Fingerprints Play in the Development of Future Treatments?
Every immune response leaves a unique signature behind, and the T cells activated by these two different pathways are no exception. Researchers have identified that T cells primed by cDC1 cells and those primed by cDC2 cells possess distinct molecular “fingerprints,” meaning they carry different biological instructions. Although both types of T cells are capable of destroying tumors, their subtle differences suggest that they might be more or less effective depending on the specific type of cancer being treated. For example, the cDC2 pathway has shown particular promise in addressing sarcomas and other connective tissue malignancies that have historically been difficult to target.
Understanding these molecular fingerprints allows for a level of precision medicine that was once considered science fiction. By analyzing a patient’s specific immune environment, clinicians can potentially tailor mRNA vaccines to favor one pathway over the other, optimizing the treatment for the individual’s needs. This insight is particularly valuable for patients who have undergone treatments like chemotherapy, which can deplete certain immune cell populations. If a patient’s cDC1 levels are low, a vaccine designed to leverage the cDC2 pathway ensures that the therapy remains a viable and powerful option for their recovery.
Summary of Scientific Advancements in Vaccine Research
The research highlights how mRNA cancer vaccines engage an unexpectedly broad range of immune cells to achieve success. By proving that cDC2 cells are capable of priming T cells through unconventional pathways such as cross-dressing, the study explains why these vaccines are so resilient in the face of complex biological challenges. The presence of redundant pathways ensures that even if one part of the immune system is compromised, another can step in to lead the charge against malignant cells. This flexibility is a hallmark of mRNA technology and sets it apart from traditional immunotherapy approaches that often rely on a more rigid set of biological requirements.
These findings serve as a roadmap for the next generation of clinical trials focusing on melanoma, lung cancer, and bladder cancer. The ability to identify the molecular fingerprints of activated T cells provides a new metric for measuring vaccine efficacy and predicting patient outcomes. As the scientific community continues to refine these tools, the focus shifts toward optimizing the delivery and formulation of vaccines to maximize the engagement of both immune pathways. This comprehensive understanding of dendritic cell coordination provides the necessary data to move beyond a general treatment model toward a truly personalized approach to oncology.
Conclusion: The Road Ahead for mRNA Immunotherapy
The revelation of dual immune pathways transformed the understanding of how the body responded to therapeutic instructions. It was clear that the redundant nature of the immune system provided a safety net that scientists were finally able to map and manipulate for better clinical results. This shift in perspective allowed for the development of more sophisticated vaccines that did not just hope for a response but actively engineered one through multiple biological channels. The study of these mechanisms proved that the effectiveness of mRNA technology was rooted in its ability to cooperate with the natural versatility of human biology.
Moving forward, the focus must remain on the practical application of these molecular insights to improve patient survival rates. Researchers and clinicians should consider how the local environment at the injection site influenced the “cross-dressing” process and whether specific adjuvants could further boost cDC2 activation. By integrating these findings into standardized treatment protocols, the medical field moved closer to a future where cancer was managed with the same precision as a targeted viral infection. The journey of mRNA from a pandemic solution to a cancer-fighting powerhouse demonstrated the enduring value of exploring the fundamental mysteries of the human immune system.