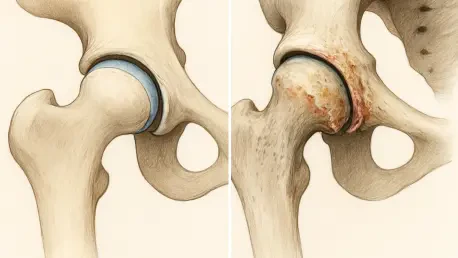
The ability to maintain mobility into senior years often depends on the structural integrity established during the very first stages of human development. A landmark multiethnic study has recently unveiled the complex genetic architecture connecting developmental dysplasia of the hip, commonly known as DDH, to the eventual onset of hip osteoarthritis. Led by researchers from Kyushu University and the RIKEN Center for Integrative Medical Sciences, this global collaboration represents a major breakthrough in orthopedic science that bridges the gap between pediatric structural issues and geriatric joint degradation. By analyzing the biological transition from initial joint instability to full-scale cartilage failure, the research provides a clearer understanding of how early-life structural flaws dictate long-term mobility. This investigation is particularly significant because it moves beyond simple mechanical observations to explore the molecular pathways that drive chronic pain and joint replacement surgery.
A Massive Global Analysis of Genetic Architecture
To solve this intricate genetic puzzle, the research team conducted an unprecedented genome-wide association study involving over 350,000 tissue samples from Japan, the United Kingdom, and across Europe. This multiethnic approach was designed to ensure the findings reflected universal biological mechanisms rather than traits specific to a single population or demographic group. By meticulously comparing individuals with shallow sockets to those with full hip dislocations, the researchers were able to pinpoint the exact genetic variations that increase susceptibility to joint failure. The scale of this meta-analysis allowed the team to identify subtle genetic signals that smaller, localized studies had previously missed. Such a comprehensive data set provides a robust foundation for understanding how polygenic traits influence the mechanical stability of the hip joint. This methodology marks a shift toward large-scale data synthesis in orthopedics, highlighting the importance of international cooperation.
The study specifically highlighted three genes, identified as COL11A2, CALN1, and TRPM7, which act as the primary biological links between initial hip instability and subsequent osteoarthritis. These genes are vital for the body’s structural integrity and cellular maintenance; for instance, COL11A2 facilitates the production of Type XI collagen, which serves as the essential scaffolding for joint cartilage. Meanwhile, CALN1 and TRPM7 regulate the calcium and magnesium signaling necessary for bone cell communication and effective tissue repair. When these specific genetic instructions are compromised, the hip joint is effectively born with structural flaws and lacks the internal cellular machinery required to repair the damage caused by daily mechanical stress. This discovery provides a concrete molecular explanation for why some individuals experience rapid joint degradation while others do not. By identifying these specific loci, scientists can now trace the path from a genetic variation to a physical deformity that requires surgical intervention.
These findings provide a compelling biological narrative for why certain patients experience rapid joint degradation despite early medical interventions or physical therapy. Essentially, the research suggests that individuals with these genetic markers are dealing with a “double hit” scenario: a joint that is structurally disadvantaged from birth and a biological system that is inherently less efficient at maintaining cartilage and bone health. This molecular insight explains the direct progression from the initial instability of DDH to the painful, chronic inflammation that characterizes hip OA. Understanding this link is crucial for developing therapies that address the underlying biological weakness rather than just the mechanical symptom. Furthermore, this perspective shifts the focus of treatment from simply correcting bone alignment to supporting the cellular environment of the joint. By recognizing the metabolic deficiencies inherent in these genetic profiles, clinicians can better tailor long-term care strategies to the specific needs of the patient.
Distinguishing Between Dysplasia and Dislocation
One of the most significant contributions of this research is the discovery that hip dysplasia and hip dislocation are not genetically identical conditions, despite their clinical similarities. While the two states share much of the same polygenic foundation, the study identified unique genetic signals that determine the specific severity of the joint abnormality. This suggests that a specific subset of genetic variations acts as a biological “tipping point,” deciding whether a joint remains merely shallow or progresses to a full dislocation during development. Such a distinction is vital for future diagnostics, as it could eventually help doctors predict clinical outcomes more accurately during infancy. If a genetic profile indicates a high risk for full dislocation, medical teams can implement more aggressive stabilization techniques immediately. This level of predictive capability represents a new frontier in pediatric orthopedics, where genetic screening complements traditional physical examinations and imaging.
The research also looked beyond standard protein-coding genes to explore the significant role of non-coding DNA and bone remodeling pathways in joint health. They found that variations in regulatory regions of the genome, which were once thought to be “junk DNA,” actually function as critical switches that control bone-forming genes. In patients with hip OA, these regulatory switches often trigger hyper-active bone remodeling, leading to the painful bone spurs and structural changes that characterize late-stage disease. This further cements the link between early genetic predispositions and the debilitating symptoms that appear later in life. By understanding how these non-coding regions influence gene expression, researchers can begin to look for ways to “turn off” the signals that lead to bone spurs. This discovery highlights the complexity of the human genome and the necessity of looking at the entire genetic landscape to understand disease. It opens up new avenues for research into how environmental factors might interact with these switches.
Pioneering Precision Medicine and Future Treatments
The clinical implications of these findings are particularly profound for populations where hip osteoarthritis is frequently rooted in developmental dysplasia, such as in Japan. By identifying these specific susceptibility loci, the medical community is moving toward a precision medicine model for orthopedics that prioritizes individualized risk assessment. This shift allows for the identification of high-risk individuals long before they require invasive procedures, opening the door for proactive management rather than reactive surgery. Instead of waiting for a patient to present with chronic pain in their fifties, doctors could theoretically use genetic data to begin protective measures in their twenties. Such measures might include targeted exercise programs, weight management, or specific nutritional support designed to bolster cartilage health. This proactive approach has the potential to significantly reduce the global burden of hip replacement surgeries and improve the overall quality of life for millions of people worldwide.
Looking ahead, researchers aim to utilize “multi-omics” to explore how these genes interact with other biological systems, such as how DNA is folded within cartilage cells. By focusing on chondrocytes, the cells responsible for cartilage health, the goal was to develop targeted pharmaceutical interventions that could bolster bone remodeling or protect joint tissue. This research path led to a better understanding of how chromatin accessibility affects the way our bodies respond to joint stress. The study suggested that early genetic screening could become a standard part of pediatric care, allowing for specialized treatments that preserve joint function throughout a patient’s life. Moving forward, the medical community focused on integrating these genetic insights into daily clinical practice, ensuring that the transition from discovery to treatment was seamless. These advancements provided a roadmap for tackling other complex musculoskeletal disorders through a combination of genetic engineering and personalized orthopedic care.