
In a significant move, Compass Pathways has announced its decision to lay off 30% of its workforce, streamline its research efforts, and explore selling its digital technologies. This decision comes in response to delays in the development of its lead drug, a synthetic form of psilocybin aimed at

Jade Biosciences, a burgeoning developer specializing in immune disease drugs, has set its sights on accelerating its market presence through a reverse merger with Aerovate Therapeutics. This strategic consolidation will enable Jade to promptly enter the public market under the Nasdaq ticker

Neurogene, a trailblazer in the gene therapy field concentrating on rare neurological diseases, has successfully clinched $200 million through a private funding deal designed to extend its financial runway until late 2027. This financing structure, recognized as a PIPE financing (Private Investment
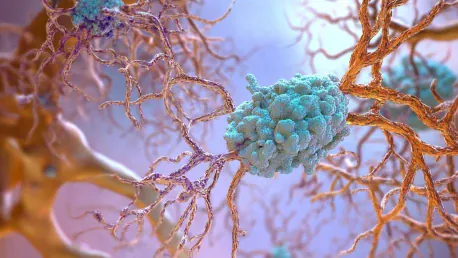
Roche is considering seeking accelerated approval for its experimental Alzheimer’s drug, trontinemab, following promising early results. Trontinemab is currently in Phase 2 testing and has shown significant potential in reducing amyloid levels in the brain. This approach has been previously
Novo Nordisk recently announced its intention to seek regulatory approval for the use of semaglutide, the active ingredient in its profitable weight loss drug, for the treatment of metabolic dysfunction-associated steatohepatitis (MASH). This decision came after a Phase 3 clinical trial yielded
In a significant development, Monte Rosa Therapeutics, a biotechnology company based in Boston, has finalized a highly lucrative global exclusive development and commercialization license agreement with Novartis valued at up to $2.1 billion on October 28, 2024. Monte Rosa Therapeutics is focused on