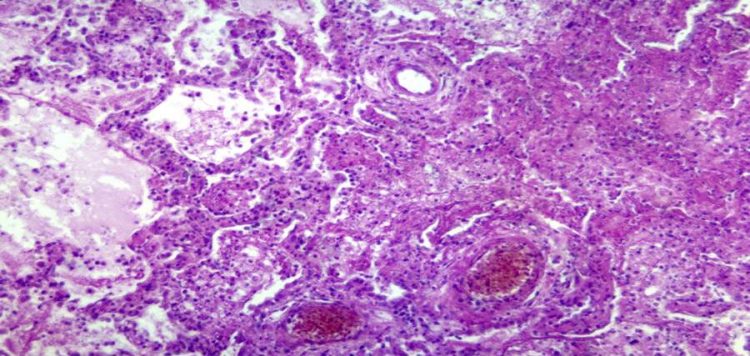
Moleculin Biotech, Inc., (Nasdaq: MBRX) (“Moleculin” or the “Company”), a clinical stage pharmaceutical company with a broad portfolio of drug candidates, today announced that it is now authorized by the Polish Department of Registration of Medicinal Products known as URPL to accelerate the Phase 1 dose escalation portion of its clinical trial of Annamycin for the treatment of acute myeloid leukemia (AML).