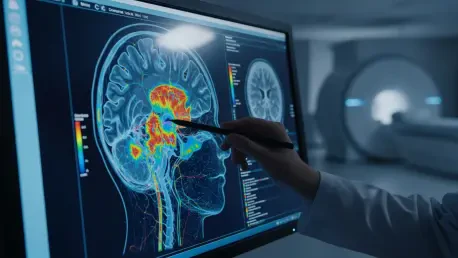
The intricate landscape of the human brain often hides the earliest whispers of neurodegeneration long before a patient ever experiences their first moment of profound or disorienting forgetfulness. Identifying these microscopic shifts has traditionally relied on clinical observation, yet a breakthrough study from Worcester Polytechnic Institute (WPI) has demonstrated that the future of diagnosis may reside in the cold, precise logic of machine learning. By utilizing advanced computational models, researchers achieved a staggering 92.87% accuracy rate in predicting Alzheimer’s disease through neuroanatomical analysis. This leap toward algorithmic intervention suggests that the subtle boundaries between natural aging and cognitive pathology are finally becoming visible to the digital eye.
This milestone is particularly significant because it transforms the diagnostic process from a subjective art into a data-driven science. For years, the medical community struggled to differentiate the early stages of cognitive decline from the standard wear and tear of a long life. The WPI study proved that by focusing on the physical structure of the brain rather than just behavioral symptoms, clinicians could theoretically intervene years before the most devastating effects of the disease take hold. This technological evolution marks a critical turning point in the global effort to manage an aging population.
The Digital Frontier of Neurological Diagnosis
Beyond the traditional confines of a doctor’s office, these algorithms acted as a powerful magnifying glass for the central nervous system. The research highlighted a paradigm shift where neurology and data science intersected to reveal structural “fingerprints” of decay. By processing information at a scale impossible for human clinicians, these machines identified patterns that might otherwise remain obscured by the noise of individual biological variation. This method moved the focus away from general memory tests toward a rigorous analysis of physical brain density and volume.
The analytical power of machine learning allows for the detection of minute changes in neuroanatomy that signal the transition from health to impairment. While a human radiologist might note significant shrinkage, an algorithm can quantify the loss of cubic millimeters across nearly a hundred different regions simultaneously. This level of granularity ensures that the diagnosis is not based on a single failing, but on a comprehensive map of the brain’s overall structural integrity.
The Urgency of Early Detection in an Aging World
As global life expectancy continues to climb, the prevalence of neurodegenerative conditions represents a mounting pressure on healthcare systems and family structures. Alzheimer’s disease often undergoes a “silent” phase, where brain tissue atrophies in secret while the individual appears fully functional. Bridging the gap between the onset of physical changes and the appearance of memory loss is essential for the efficacy of emerging treatments and preventative lifestyle changes. Early detection provides the only window of opportunity for patients to modify their health trajectories.
Identifying these physical markers early addresses a critical need for objective, high-precision diagnostic tools. Current clinical assessments are often reactive, occurring only after the patient or their family notices significant cognitive gaps. By the time these symptoms are obvious, the damage to the brain is frequently extensive. Moving the diagnostic timeline forward through technology allows for a proactive stance, where pharmaceutical and behavioral interventions can be applied when the brain still possesses a degree of resilience.
Decoding the WPI Study: Algorithms and Anatomy
The success of this 93% accuracy claim rested on a sophisticated two-step computational strategy applied to a massive dataset. Researchers utilized 815 high-resolution MRI scans from the Alzheimer’s Disease Neuroimaging Initiative, covering a spectrum from healthy individuals to those with confirmed Alzheimer’s. The first phase involved a specialized algorithm designed to isolate and measure the volume of 95 distinct brain regions, providing a granular look at the physical architecture of each participant’s brain. This automated mapping removed the risk of human error and provided a standardized baseline for comparison.
A second machine learning model then analyzed these volumetric measurements to identify specific patterns of atrophy. This allowed the system to distinguish the structural signatures of Alzheimer’s from other forms of cognitive decline or natural aging. The study pinpointed the hippocampus, amygdala, and entorhinal cortex as the most significant predictors of the disease. These regions are central to memory and emotion, and their specific rate of decline became the “smoking gun” that the algorithm used to reach its high level of certainty.
Expert Insights into Gender and Age-Based Vulnerabilities
Leading researchers, including Assistant Research Professor Benjamin Nephew, emphasized that Alzheimer’s does not manifest identically across all demographics. The study revealed that while certain markers are universal, others are highly dependent on biological sex. For instance, women showed more pronounced volume loss in the left middle temporal cortex, while men exhibited significant atrophy in the right entorhinal cortex. These variations suggested that a personalized approach, accounting for hormonal differences such as estrogen and testosterone, would be necessary for future clinical success.
The research team also noted the generalizability of their findings, suggesting that these biomarkers represent universal indicators rather than study-specific anomalies. By understanding how the disease progresses differently in men and women, developers can refine diagnostic software to weight specific brain regions differently based on the patient’s profile. This nuance ensures that the 93% accuracy rate remains consistent across a diverse global population, moving away from a “one-size-fits-all” model that often fails to account for sex-based biological realities.
Integrating Predictive Tech into Clinical Frameworks
The integration of these predictive technologies into standard clinical frameworks signaled the dawn of a more proactive era in neuro-diagnostics. Researchers recognized that transitioning from subjective cognitive tests to standardized volumetric analysis of the hippocampus and entorhinal cortex was a vital step for practitioners. They determined that future software needed to weight specific brain regions differently based on sex and age to maintain a high degree of precision. This transition required a shift in how medical facilities prioritized imaging data and computational resources.
Interdisciplinary synergy between computer scientists and neurologists became the primary driver for refining deep learning models that accounted for comorbidities like diabetes. This collaborative environment ensured that brain atrophy was no longer viewed in isolation but as part of a complex, systemic health profile. The successful application of these algorithms meant that the medical field moved closer to a reality where neurodegeneration was managed through data-driven analysis. Ultimately, the focus shifted toward developing accessible software interfaces that allowed local clinics to upload MRI data and receive high-probability risk assessments in real time.