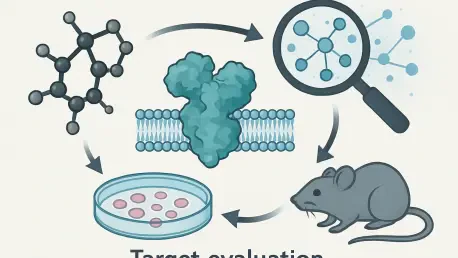
In the fast-evolving world of biopharmaceuticals, identifying the right drug discovery target remains a cornerstone of successful research and development, often determining whether a project thrives or falters in its early stages. The complexity of this decision-making process, coupled with the
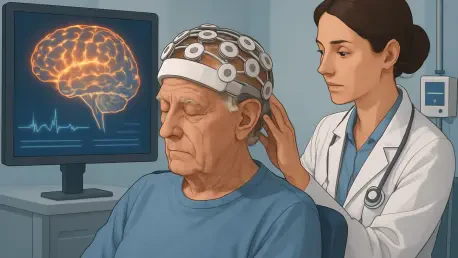
The world stands at a critical juncture as Alzheimer’s disease (AD), the predominant cause of dementia, affects over 40 million individuals globally, with projections estimating a staggering rise to 140-150 million cases by 2050, creating an urgent need for innovative solutions. This relentless
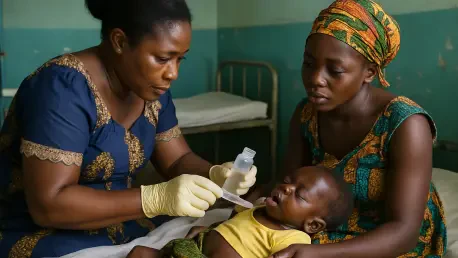
In the heart of West Africa, Ghana faces a relentless battle against malaria, a disease that continues to claim the lives of countless infants each year due to their vulnerable immune systems and the historical absence of treatments designed specifically for them, leaving families desperate for
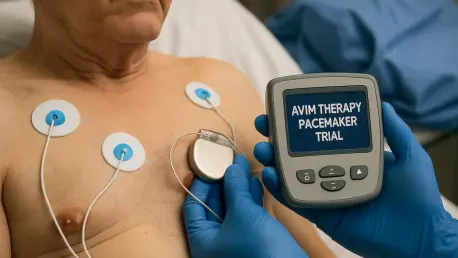
What if a small device in millions of chests across America could do more than regulate heartbeats—what if it could also tame the silent threat of high blood pressure? For countless older adults with pacemakers, uncontrolled hypertension remains a persistent danger, often resistant to standard

What if the key to defeating devastating diseases like cancer or antibiotic-resistant infections could be unlocked in mere days instead of months or years, transforming the landscape of medical research? This isn't a distant dream but a tangible reality with a groundbreaking tool developed by
Imagine a world where damaged tissues can be regenerated with pinpoint accuracy, where therapies are tailored to an individual’s unique genetic makeup, and where neurological disorders are treated by targeting the exact molecular missteps. This vision is becoming a reality thanks to a revolutionary