The human brain possesses an intricate waste-clearance system that, when functioning correctly, prevents the toxic buildup of proteins associated with cognitive decline. However, for millions of individuals, this biological janitorial service fails, leading to the relentless progression of Alzheimer’s disease. Traditional medicine has long relied on external infusions of monoclonal antibodies to fill this gap, but these treatments require lifelong maintenance and often struggle with the blood-brain barrier. A transformative shift is occurring as researchers pivot toward engineering the brain’s own resident cells to perform these tasks autonomously.
This review examines the emergence of CAR-astrocyte technology, a sophisticated leap in cellular immunotherapy. By repurposing the Chimeric Antigen Receptor (CAR) platform—originally perfected for liquid cancers—scientists have managed to turn common brain cells into proactive defenders. This approach does not just supplement the immune system; it fundamentally rewrites the functional identity of the central nervous system’s most abundant cells, offering a localized and potentially permanent solution to neurodegeneration.
Evolution of Cellular Immunotherapy for Neurodegenerative Disorders
The journey toward effective Alzheimer’s treatment has transitioned from simple symptom management to aggressive plaque removal. Historically, the industry focused on monoclonal antibodies that target amyloid beta. While effective at slowing decline, these therapies are hindered by their delivery requirements, necessitating frequent, high-dose intravenous infusions. This creates a “peak-and-trough” effect where the drug concentration in the brain is rarely optimal, and the logistical burden on patients remains immense.
The move toward CAR-astrocyte therapy represents a departure from these transient pharmacological interventions. Instead of introducing foreign proteins that the body eventually clears, this technology utilizes viral vectors to deliver genetic instructions directly to the brain. This creates a self-sustaining population of modified cells. It marks a critical evolution from “treating” a disease with external chemicals to “curing” it through internal biological reprogramming, effectively establishing a living pharmacy within the cranium.
Engineering the CAR-Astrocyte System
Reprogramming Astrocytes into Biological Super-Cleaners
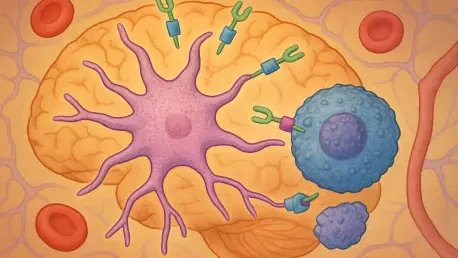
At the heart of this innovation is the astrocyte, a star-shaped glial cell traditionally viewed as a supporting character to neurons. Under normal conditions, these cells maintain the chemical environment of the brain but lack the aggressive phagocytic (eating) capabilities of professional immune cells like microglia. By inserting a specific genetic sequence via a viral delivery system, engineers can “unlock” a latent potential within these cells, essentially transforming them into biological super-cleaners.
This reprogramming is significant because astrocytes are far more numerous and stable than microglia. While microglia often become inflamed or dysfunctional during the progression of Alzheimer’s, reprogrammed astrocytes remain resilient. They provide a massive, distributed network of waste-processing units that can handle a much higher volume of amyloid beta. Performance metrics from early trials indicate that these engineered cells can sustain their cleaning activities for months following a single intervention, a feat impossible for standard drug molecules.
The Chimeric Antigen Receptor (CAR) Homing Mechanism
The precision of this therapy is dictated by the Chimeric Antigen Receptor, or CAR. In this system, the CAR acts as a highly specific biological GPS. It consists of an extracellular domain that recognizes the unique signature of amyloid beta plaques and an intracellular domain that triggers the astrocyte to engulf and digest the target. This mechanism ensures that the cells do not attack healthy brain tissue, providing a level of specificity that traditional drugs often lack.
Unlike antibodies that circulate blindly through the bloodstream, the CAR-astrocyte system is localized. Once the astrocytes are modified, they stay in place, monitoring their immediate environment for the first signs of plaque formation. This constant surveillance allows for real-time response to protein misfolding. In real-world usage scenarios within laboratory models, this has resulted in a 50% reduction in existing plaque volume, demonstrating that the CAR mechanism is not just a preventative tool but a powerful corrective one.
Current Trends in Brain Waste Clearance and Neuro-Immunology
The field of neuro-immunology is currently moving away from systemic treatments in favor of “precision-guided” cellular therapies. There is an increasing realization that the brain’s unique environment requires a tailored approach that respects the blood-brain barrier rather than trying to force its way through it. Industry behavior is shifting toward “one-and-done” genetic therapies that reduce the long-term cost of care and minimize the side effects associated with high-dose systemic drugs.
Moreover, there is a growing trend of “cell-type diversification” in research. While T-cells were the primary focus of immunotherapy for years, the successful recruitment of astrocytes indicates that the next generation of medicine will utilize the most relevant cell for the specific organ being treated. This shift reflects a broader technological trajectory toward decentralized, organ-specific immune responses that operate independently of the body’s general lymphatic system.
Real-World Applications in Chronic Disease Management
Beyond the immediate scope of Alzheimer’s, the deployment of CAR-astrocytes has profound implications for a variety of chronic neurological conditions. For instance, in diseases like Parkinson’s or ALS, where specific toxic proteins aggregate and destroy neurons, the CAR system could be recalibrated to target alpha-synuclein or TDP-43. This modularity makes the technology a versatile platform for the entire spectrum of neurodegenerative disorders, allowing for custom-designed “cleaner” cells based on a patient’s specific protein pathology.
In the oncology sector, this technology is being adapted to fight aggressive brain tumors like glioblastoma. By engineering astrocytes to recognize malignant cell markers, researchers are creating a resident defense force that can attack tumors from within the brain tissue. This implementation is particularly notable because it bypasses the traditional limitations of chemotherapy, which often fails to reach effective concentrations in the brain without causing severe systemic toxicity.
Technical Hurdles and Clinical Implementation Challenges
Despite its promise, the transition from lab to clinic faces significant technical hurdles. One primary concern is the potential for “off-target” effects. If an engineered astrocyte were to misidentify a healthy protein as a threat, it could lead to unintended neuroinflammation or the destruction of essential synapses. Controlling the “intensity” of the astrocyte’s cleaning response is vital to ensure that the therapy does not become as damaging as the disease it seeks to treat.
Regulatory and market obstacles also persist. Traditional drug approval processes are designed for repeatable doses of chemical compounds, not single-dose genetic modifications that persist for years. There are also concerns regarding the delivery method; using viral vectors requires precise neurosurgical techniques that may limit the therapy’s availability to specialized medical centers. Ongoing development is currently focused on non-invasive delivery methods, such as ultrasound-mediated blood-brain barrier opening, to make the treatment more accessible.
Future Prospects: From Alzheimer’s to Oncology
The long-term impact of CAR-astrocyte technology will likely be measured by its ability to move toward prophylactic use. In the future, individuals with a high genetic predisposition for dementia might receive a single “guardian” injection in mid-life, effectively immunizing their brain against protein accumulation before symptoms ever appear. This would represent a total paradigm shift from reactive medicine to proactive biological fortification.
Furthermore, the integration of synthetic biology could lead to “switchable” CAR-astrocytes. These cells could be programmed to remain dormant until they receive a specific chemical signal or until they detect a certain threshold of pathology. This level of control would mitigate many of the safety concerns currently associated with permanent genetic changes, allowing clinicians to turn the therapy on or off as needed. The potential for these cells to act as both sensors and responders positions them as the cornerstone of future neuro-restorative medicine.
Assessment of the Shift Toward One-Time Biological Interventions
The emergence of CAR-astrocyte therapy proved that the most effective way to manage complex neurological diseases was to empower the brain’s internal architecture rather than overwhelming it with external chemicals. By successfully merging the specificity of cancer immunotherapy with the abundance of glial cells, researchers provided a blueprint for long-term disease modification that surpassed the efficacy of traditional monoclonal antibodies. This transition signaled a departure from the era of chronic pharmaceutical dependence, moving the industry toward a model of definitive biological correction.
The shift was not merely a technical success but a fundamental change in how society approached chronic illness. By focusing on a single-dose intervention, the medical community addressed the logistical and financial burdens that previously made advanced Alzheimer’s care inaccessible to many. Ultimately, the development of these engineered “super-cleaners” established a new standard for precision medicine, demonstrating that with the right genetic instructions, the body could be taught to heal itself from within.