
The pharmaceutical landscape often operates on a razor's edge where multi-billion dollar investments can be validated or neutralized by a single data readout from a Phase 3 clinical trial. Merck and Gilead Sciences currently find themselves navigating this volatility as they balance the demands of

As a domestic feline transitions from the cushioned comfort of a living room sofa to the unmonitored shadows of a backyard thicket, it effectively functions as a mobile biological sensor that bridges two vastly different ecological zones. This unique mobility makes domestic cats a critical subject

The traditional image of healthcare as a monolith of sprawling hospital campuses and cold clinical environments is rapidly dissolving in the face of a decentralized digital movement. As the global telehealth market accelerates toward a staggering four hundred and fifty billion dollar valuation by

The industrial landscape of the American Midwest is undergoing a profound transformation as Ohio emerges as a primary center for advanced pharmaceutical production, fueled by a substantial $267 million investment from Hikma Pharmaceuticals. This massive infusion of capital serves as a cornerstone

The persistent struggle against KRAS mutations has long been a defining battle in modern oncology, representing a target once thought to be totally unreachable by conventional drug design due to the protein's lack of accessible binding pockets. For decades, clinicians and researchers faced a
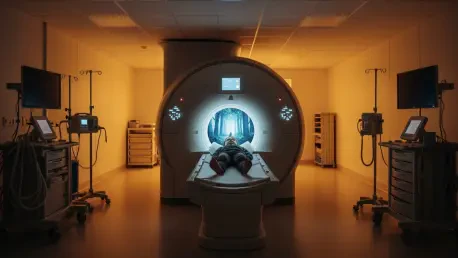
For a child, the cavernous bore of an MRI machine, coupled with its rhythmic thumping and the necessity for absolute stillness, can be a terrifying experience that often results in failed scans or the need for general anesthesia. This fundamental challenge in pediatric radiology has prompted a