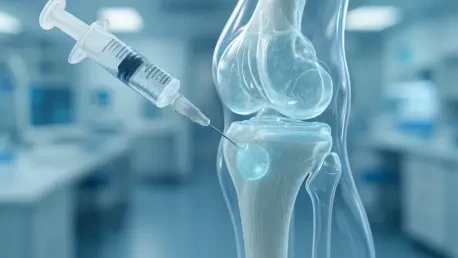
The persistent struggle against degenerative joint diseases has reached a critical turning point as medical researchers move away from temporary pain management toward true biological restoration. For decades, the medical community viewed the erosion of cartilage as an inevitable consequence of aging or injury, leaving patients with limited options that often culminated in invasive and costly joint replacement surgeries. Osteoarthritis currently affects hundreds of millions of individuals globally, serving as a primary driver of chronic disability and decreased quality of life. This condition involves the progressive breakdown of joint tissues, where the protective cartilage that cushions the ends of bones wears thin, eventually leading to painful bone-on-bone contact. Until now, the primary clinical objective was simply to slow the decline or mask the discomfort. However, a revolutionary injectable therapy is currently redefining expectations by focusing on actual tissue reversal and regeneration.
A Biological Shift: Engineering the Body to Heal Itself
Central to this breakthrough is a sophisticated, slow-release drug-delivery system designed to communicate directly with the body’s innate biological machinery. Developed by a multidisciplinary team led by chemical and biological engineer Stephanie Bryant at the University of Colorado Boulder and UC Anschutz, this technology functions by coaxing existing cartilage and bone cells to initiate a repair sequence. Instead of merely introducing a lubricant or a steroid, the injection delivers specific biochemical cues that trigger the natural regeneration of the extracellular matrix. In preliminary animal studies, this approach demonstrated a remarkable ability to reverse established damage within a matter of weeks, proving that the body maintains the potential to heal even supposedly permanent structural decays. The speed of this development has been exceptional, moving from a conceptual moonshot to a functional therapy in roughly two years through intense collaboration.
Beyond the liquid delivery of regenerative signals, the research team is also refining an injectable hydrogel scaffold that acts as a temporary physical implant. This material is engineered to set in place within the joint, filling the physical gaps and fissures that characterize advanced osteoarthritis. Once situated, the scaffold serves as a recruitment center, drawing in healthy cells from surrounding tissues to populate the damaged area and create a new, durable surface. This dual-action approach allows for tailored interventions based on the specific severity of a patient’s condition, addressing everything from early-stage wear to more significant structural voids. By providing a structural framework that eventually dissolves as it is replaced by living tissue, this method avoids the long-term complications associated with permanent synthetic implants. It represents a shift from mechanical replacement to biological architecture, ensuring that the new joint surface is healthy.
Clinical Pathways: The Journey Toward Widespread Implementation
As this technology moves into its second phase, the focus has shifted toward establishing rigorous safety and toxicology profiles necessary for human application. This critical stage is supported by the Novel Innovations for Tissue Regeneration in Osteoarthritis program, an initiative under the federal Advanced Research Projects Agency for Health, which aims to accelerate high-impact medical solutions. Current projections indicate that human clinical trials are slated to begin within the next 18 months, marking a significant milestone in the transition from laboratory success to real-world clinical availability. The goal of this federal backing is to provide a viable alternative to the cycle of repeat surgeries and chronic medication use that currently burdens the healthcare system. By prioritizing therapies that can be administered in a simple outpatient setting, researchers hope to democratize access to advanced medicine, ensuring that restoration becomes as routine as a minor procedure.
This progression toward regenerative solutions occurred alongside a growing scientific consensus regarding the importance of proactive joint health and early intervention. While researchers at Stanford University identified specific proteins linked to cartilage loss, others investigated the potential role of metabolic medications like semaglutide in supporting joint integrity. These diverse efforts converged into a unified strategy that viewed osteoarthritis not as a permanent disability, but as a manageable and curable pathology. Patients and clinicians alike recognized that the key to long-term mobility resided in the body’s own regenerative capacity rather than in mechanical substitutes. This paradigm shift encouraged a new focus on monitoring joint health biomarkers long before structural failure occurred. Consequently, the medical community successfully moved toward a future where maintaining independence and high activity levels became the standard expectation for the aging population.