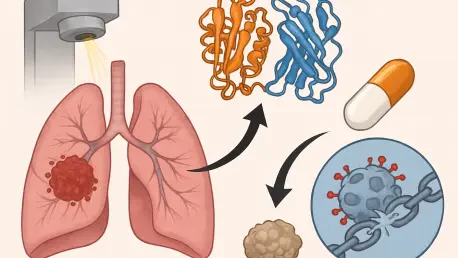
The persistent struggle against lung cancer has long relied on radiation therapy to dismantle the genetic integrity of malignant cells, yet many aggressive tumors possess an uncanny ability to withstand these high-energy assaults. Recent breakthroughs from The University of Texas MD Anderson Cancer Center have pinpointed a specific metabolic culprit: the mitochondrial enzyme dihydroorotate dehydrogenase, commonly known as DHODH. This enzyme acts as a sophisticated internal shield, allowing cancer cells to neutralize the oxidative damage that would normally trigger programmed cell death. By identifying this specific vulnerability, researchers are moving toward a new era where the biological defenses of a tumor can be systematically dismantled, ensuring that radiation therapy achieves its intended lethal effect. This discovery is particularly vital because it explains why some patients experience rapid recurrence despite receiving the maximum tolerated doses of conventional radiotherapy, providing a clear target for future pharmacological intervention and improved clinical outcomes.
The fundamental mechanism by which radiation eliminates cancer cells is through a process called ferroptosis, which is an iron-dependent form of cell death characterized by the massive accumulation of lipid peroxides. Under normal conditions, radiation-induced stress overwhelms the cell’s antioxidant capacity, leading to structural collapse. However, lung cancer cells frequently upregulate DHODH activity immediately following exposure to radiation, creating a protective buffer. While DHODH is widely recognized for its role in synthesizing nucleotides necessary for DNA repair, its secondary function is arguably more critical for survival. It facilitates the production of ubiquinol, a potent antioxidant that actively scavenges the lipid radicals generated during treatment. By suppressing the chemical signals required to initiate ferroptosis, DHODH effectively grants the tumor a reprieve, allowing it to survive and eventually proliferate despite the intensive treatment administered by oncologists.
Strategic Metabolic Intervention
Repurposing Existing Medication for Oncology
The transition from laboratory discovery to clinical application is often a decade-long journey, but the identification of DHODH as a target offers a unique shortcut through the use of leflunomide. Originally approved by the FDA for the management of rheumatoid arthritis, leflunomide is a potent inhibitor of DHODH with a safety profile that has been thoroughly documented over years of widespread use. Because the pharmaceutical industry and medical community are already familiar with the side effects and metabolic behavior of this drug, the path to incorporating it into cancer treatment protocols is significantly streamlined. This repurposing strategy eliminates the need for the extensive phase-one toxicity trials that typically delay new oncology drugs, allowing researchers to focus immediately on proving its efficacy in enhancing radiation sensitivity within lung cancer patients.
Utilizing an established drug like leflunomide also addresses the economic and logistical barriers that often prevent patients from accessing cutting-edge therapies. In the current medical landscape of 2026, the cost of developing novel targeted inhibitors can be astronomical, frequently resulting in treatments that are financially out of reach for many. Leflunomide offers a cost-effective alternative that can be integrated into existing hospital workflows without requiring specialized training or complex new equipment. By disrupting the metabolic shield of the tumor with a known agent, clinicians can effectively “prime” the cancer cells for destruction. This approach ensures that the primary treatment—radiation—is no longer firing into a fortified bunker but is instead striking a vulnerable target that has lost its primary means of internal repair and antioxidant defense.
The Power of Triple Combination Therapy
While the inhibition of DHODH provides a notable advantage, preclinical models suggest that the most dramatic therapeutic results occur when this intervention is part of a coordinated triple-threat strategy. This regimen combines traditional radiation with immune checkpoint inhibitors and DHODH suppression, creating a synergistic effect that no single treatment can match. Radiation serves as the initial hammer, causing physical damage and releasing tumor antigens into the bloodstream. These antigens then act as a beacon for the immune system, which is further empowered by checkpoint blockade therapy. However, the true breakthrough lies in how leflunomide removes the metabolic “brakes” that often prevent these immune responses from being effective. By neutralizing DHODH, the treatment ensures that the pro-death signals sent by the immune system are not blocked by the tumor’s antioxidant defenses.
Specifically, the interaction between the immune system and the tumor metabolism involves a critical signaling molecule known as interferon-gamma. In a typical scenario, immunotherapy stimulates the production of this molecule, which is supposed to drive the cancer cell toward ferroptosis. Unfortunately, many radioresistant tumors use DHODH to counteract this signal, effectively ignoring the instructions to die. When leflunomide is added to the mix, this internal resistance is bypassed. The triple combination forces the cell into a corner where it cannot repair its DNA, cannot neutralize oxidative stress, and cannot ignore the immune system’s attack. This comprehensive strategy addresses the multifaceted nature of cancer resistance, ensuring that if a cell survives the initial radiation blast, it remains highly susceptible to the secondary and tertiary waves of the therapeutic onslaught.
Future Implications for Cancer Treatment
Shifting the Paradigm of Tumor Vulnerability
The shift toward targeting metabolic pathways like DHODH represents a profound evolution in how the oncology community perceives and treats malignant growth. Rather than simply escalating the physical intensity of treatments—which often leads to debilitating side effects for the patient—the focus has moved toward identifying and disabling the adaptive pathways that allow tumors to survive. This approach treats the tumor as a dynamic, evolving entity rather than a static target. By understanding the specific “druggable” vulnerabilities within the cell’s internal machinery, scientists can create personalized treatment plans that strip away a tumor’s resilience. This paradigm shift encourages a more surgical and intellectual approach to cancer care, where the goal is to outsmart the biology of the disease rather than just overwhelming it with brute force.
Furthermore, this metabolic focus provides a framework for addressing the problem of acquired resistance, which remains one of the greatest hurdles in modern medicine. When a tumor evolves to survive one form of therapy, it often does so by diverting energy or resources through alternative metabolic routes. By targeting a foundational enzyme like DHODH, which is essential for both DNA synthesis and antioxidant protection, clinicians can close off multiple survival avenues simultaneously. This reduces the likelihood that the cancer will find a “workaround” to the treatment. As we progress from 2026 to 2028, the integration of metabolic inhibitors into standard oncology protocols is expected to become a primary focus, transforming what were once considered untreatable, radioresistant cases into manageable conditions that respond predictably to combined therapeutic interventions.
Broad Applications Across Solid Tumors
The discovery that DHODH inhibition can reverse radiation resistance in lung cancer has implications that ripple across the entire field of solid tumor research. Because the biological processes of ferroptosis and oxidative stress management are nearly universal across different types of cancer, the triple-threat regimen could potentially be applied to breast, prostate, and pancreatic cancers. Many of these malignancies also exhibit a stubborn resistance to radiation, often through similar metabolic shielding techniques. If the clinical trials currently being designed can replicate the success seen in preclinical models, this strategy will offer a robust, evidence-based pathway for treating a wide array of hard-to-manage diseases. The convergence of metabolism, immunology, and radiotherapy provides a versatile toolkit that can be adjusted based on the specific metabolic profile of any given tumor.
Looking forward, the success of this research highlights the necessity of multi-modal treatment strategies that address the complexity of cancer on several biological levels at once. Future clinical trials must focus on identifying the optimal timing for administering DHODH inhibitors relative to radiation and immunotherapy to maximize the ferroptotic response. Additionally, the development of biomarkers to identify patients with high DHODH activity will be essential for tailoring these treatments to those who will benefit most. By moving beyond the limitations of single-agent therapies and embracing the synergy of combined metabolic and immunological attacks, the medical community is setting the stage for a significant increase in long-term survival rates. This integrated approach ensures that the next generation of cancer care is not only more effective but also more precise, directly addressing the underlying mechanisms that have historically made cancer so difficult to eradicate.