The high-stakes world of oncology has long viewed triple-negative breast cancer as a fortress that resists the most sophisticated modern weapons, leaving patients and physicians with few options beyond traditional chemotherapy. This aggressive subtype, notorious for its rapid progression and lack of targeted receptors, has historically forced a reliance on toxic systemic treatments that often yield diminishing returns. However, the medical community is currently buzzing with the announcement of the BL-B01D1-307 trial results, which showcase a first-in-class bispecific antibody-drug conjugate known as izalontamab brengitecan, or iza-bren. For the first time in the history of this specific patient population, a therapeutic agent has achieved a statistically significant improvement in both progression-free and overall survival compared to standard chemotherapy, marking a monumental shift in the standard of care.
A Landmark Shift in the Fight Against Aggressive Malignancies
For decades, the standard response to a diagnosis of triple-negative breast cancer (TNBC) was a sobering acknowledgement of the limitations of modern medicine. While other forms of breast cancer benefited from the revolution in hormone therapies and HER2-targeted drugs, TNBC remained a stubborn outlier. The recent data regarding izalontamab brengitecan represents more than just a successful clinical trial; it signifies the breaching of a biological wall that once seemed insurmountable. Researchers have finally identified a way to deliver potent anticancer agents directly into the heart of these tumors with a precision that was previously impossible.
The clinical success of iza-bren serves as a validation for the next generation of antibody-drug conjugates (ADCs). By moving beyond single-target molecules, scientists have unlocked a method to address the heterogeneity of aggressive tumors. This breakthrough offers a sense of renewed purpose for oncology departments worldwide, as it suggests that even the most recalcitrant cancers have vulnerabilities that can be exploited through clever engineering. The shift from broad-spectrum exhaustion toward targeted molecular destruction is now a tangible reality for patients who previously had little reason for optimism.
The Clinical Challenge of Triple-Negative Breast Cancer
The urgency surrounding the development of iza-bren is rooted in the biological “invisibility” of TNBC. By definition, this cancer lacks estrogen, progesterone, and HER2 receptors, which effectively renders it immune to the blockbuster drugs that have transformed other breast cancer outcomes. Because these traditional docking stations are absent, the cancer cells often proliferate unchecked, frequently migrating to the lungs, liver, or brain. When a patient progresses after initial taxane-based chemotherapy, the medical road often hits a dead end, leaving few late-line alternatives that can actually extend life rather than just managing symptoms.
Furthermore, the social and emotional toll of a TNBC diagnosis is compounded by the speed at which the disease moves. It is often diagnosed in younger patients and carries a higher risk of recurrence within the first few years following treatment. For those living with unresectable locally advanced or metastatic disease, the hunt for a therapy that can bypass traditional resistance mechanisms is a race against time. The medical community has spent years searching for a “Trojan Horse” capable of entering these cells without needing the three classic receptors, and the emergence of bispecific targeting finally addresses this critical necessity for survival.
Understanding the Innovation: How Izalontamab Brengitecan Works
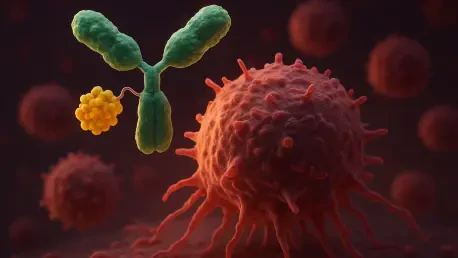
The engineering behind izalontamab brengitecan is a masterclass in molecular precision, utilizing a bispecific targeting system that hunts for two distinct proteins: EGFR and HER3. While many cancers might lose or downregulate a single receptor to evade treatment, iza-bren’s dual-action approach makes it much harder for the tumor to hide. EGFR and HER3 are frequently overexpressed in epithelial cancers and are notorious for driving the signaling pathways that help tumors survive and develop resistance to conventional drugs. By locking onto both, the drug ensures a higher degree of specificity and a stronger bond to the malignant cell.
Once the drug binds to these dual receptors, it is internalized through a process that draws the entire complex inside the cancer cell. At this point, the “Trojan Horse” releases its deadly cargo—a potent topoisomerase 1 inhibitor payload. This specific chemical agent is designed to disrupt the DNA replication process within the tumor, causing the cell to trigger its own destruction. This localized release means that the toxic effects are concentrated where they are needed most, significantly reducing the collateral damage to healthy tissues that often makes traditional chemotherapy so difficult for patients to endure.
Evidence from the BL-B01D1-307 Phase III Trial
The validation for this innovative approach came through the rigorous BL-B01D1-307 trial, a multicenter study that pitted iza-bren against the physician’s choice of standard chemotherapy. The results were nothing short of a paradigm shift for the industry. It was the first time a bispecific ADC delivered dual positive results for both progression-free survival (PFS) and overall survival (OS) in a TNBC setting. This means that patients not only lived longer without their cancer growing, but they also lived longer overall—a gold-standard achievement that few drugs in this category have ever reached.
Experts who analyzed the topline findings noted that the superiority of iza-bren over the long-standing standard of care was consistent across various patient subgroups. The data suggested that the EGFRxHER3-targeting mechanism remained effective even in patients who had already undergone multiple rounds of prior therapy. This level of efficacy in a late-line setting provides a robust argument for the drug’s integration into earlier stages of treatment. The success of this trial has effectively moved bispecific ADCs from the realm of experimental curiosity to a primary pillar of modern oncological strategy.
Navigating the Future: Potential Applications and Global Strategy
The path forward for izalontamab brengitecan involves a complex global strategy to ensure that this breakthrough reaches the patients who need it most. Regulatory bodies in major markets began priority reviews as the data became available, with New Drug Applications moving swiftly through the approval process. The momentum generated by the TNBC results also fueled investigations into other epithelial cancers, such as non-small cell lung cancer and esophageal carcinoma. This versatility suggests that the drug could eventually become a broad-spectrum tool for a variety of difficult-to-treat solid tumors.
Strategic partnerships between companies like Biokin, SystImmune, and Bristol Myers Squibb highlighted the collaborative nature of this new era in medicine. These alliances aimed to streamline the supply chain and make the therapy accessible beyond the initial trial regions. For clinicians, the focus shifted toward evaluating treatment sequences to determine where bispecific ADCs fit best within a personalized care plan. The goal was no longer just to find a treatment that worked, but to optimize the timing and combination of these therapies to maximize long-term survival and quality of life for every patient.
The scientific community recognized that the success of iza-bren provided a blueprint for future drug development in aggressive oncology. Researchers pursued new combinations of bispecific antibodies and diverse payloads to stay ahead of cancer’s ability to mutate. Health systems worked to integrate genomic testing more deeply into routine care, ensuring that patients with high EGFR or HER3 expression were identified early in their treatment journey. Ultimately, the industry moved toward a model where the biological profile of the tumor, rather than its location in the body, dictated the choice of therapy. This shift allowed for a more proactive and precise approach to cancer care that favored long-term remission over temporary management.